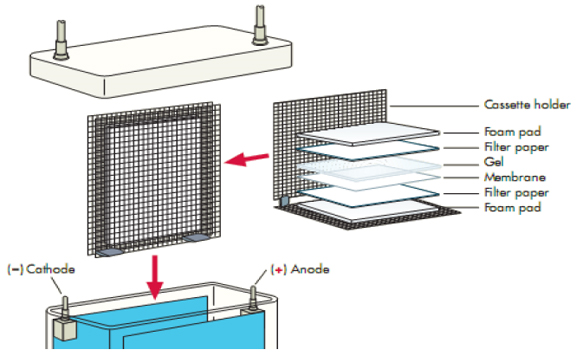
Overview of extraction options for different cells and tissuesīacteria, yeast, plant tissues, fungal cells Here we list some recommended sample extract method for several kinds of materials in Table 1. Protect the proteins to keep them from denaturation.Įxtract proteins quickly, on ice if possible and use buffer to maintain a suitable pH in order to prevent the protein degradation. Use extraction procedures that are as mild as possible. Regardless of the source and protein of interest, however, the aim must be to harvest the proteins of interest, while at the same time, obtaining a sufficient yield of proteins at an acceptable level of purity. Extraction of the proteins from plants, bacteria and fungi are further complicated. Whereas sources such as mammalian cells in suspension are easily disrupted under mild conditions and readily release their proteins, it is more difficult to extract proteins from intact tissues or within solid tumors. In principle, all sources of protein, from single cells to whole tissues as well as extracellular matrices, biological fluids and proteins secreted in vitro, are open to analysis by Western blotting. 
By understanding the nature of your starting sample and having a clear picture of the information you wish to derive from your Western blotting experiments, the chances of a successful analysis could be insured. A good sample preparation makes your western blot half success. Sample preparation is the first step and one of the most important steps of western blot. From sample preparation to protein electrophoresis. The membrane exposure time to the film/imager will depend on the abundance of the protein and the detection system.Figure 1.Note: If using a commercial kit, follow the manufacturer’s instructions. Proceed to detection using an enhanced chemiluminescence (ECL) system.Wash the membrane with Washing Buffer for 3 x 10 minutes at room temperature.Incubate the secondary antibody in PBS with 1% BSA and 0.1% Tween-20 for 1 hour at room temperature with gentle agitation.Note: If you are using a secondary antibody conjugated to HRP, do not use solutions containing NaN 3 from this point on. Wash the membrane with Washing Buffer (PBS and 0.1% Tween-20) for 3 x 10 minutes at room temperature.Incubate for 2–3 hours at room temperature or overnight at 4☌ with gentle agitation.Note: If you use a blocking peptide as a negative control, refer to our Peptide Blocking Protocol for WB. Add the primary antibody diluted in PBS 1% BSA, 0.1% Tween-20, and 0.05% NaN 3.Block the membrane with Blocking Solution (PBS with 3% BSA and 0.05% NaN 3) for 2–5 hours at room temperature with gentle agitation. 
Note: High molecular weight proteins may need a longer transfer time (see Western Blot Protocol for High Molecular Weight (HMW) Proteins ). For dry transfer, follow the manufacturer’s instructions. Transfer to a nitrocellulose membrane at 200 mA for 2.5 hours at 4☌ for wet transfer.Run the gel according to the manufacturer’s instructions.*Load 80–100 µg tissue lysate/lane or lysate from 2–5 x 10 5 cells/lane. Heat the samples in Laemmli buffer at 70–100 ☌ for 10 minutes.If you have any problems, please see our extensive troubleshooting guides. An important and widely used tool in biology, western blotting involves separating proteins according to their size via gel electrophoresis and then transferring them to a membrane to detect with specific antibodies. Subscribe – Newsletters and Email UpdatesĪ western blot (WB) lets you detect and evaluate the size of specific proteins in cell or tissue extracts.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
